In a world where infection control is more important than ever, products which have proven bactericidal efficacy are the ideal solution in healthcare facilities and other areas where hygiene cannot be compromised.
Acrovyn Bactericide pro-actively kills bacteria through surface contact alone, helping to protect patients from Healthcare-Associated Infections (also referred to as nosocomial infections). It has been tested to ISO 22196* and proven up to 100% effective against most commonly occurring hospital and food bacteria strains.
Sanitized® zinc pyrithione*, the advanced anti-microbial additive incorporated during manufacture of Acrovyn Bactericide products, remains effective throughout their lifecycle and is not affected by most stringent cleaning regimes.
The products are completely safe to humans but deadly for bacteria, guaranteeing long term hygienic conditions.
Acrovyn Bactericide Products
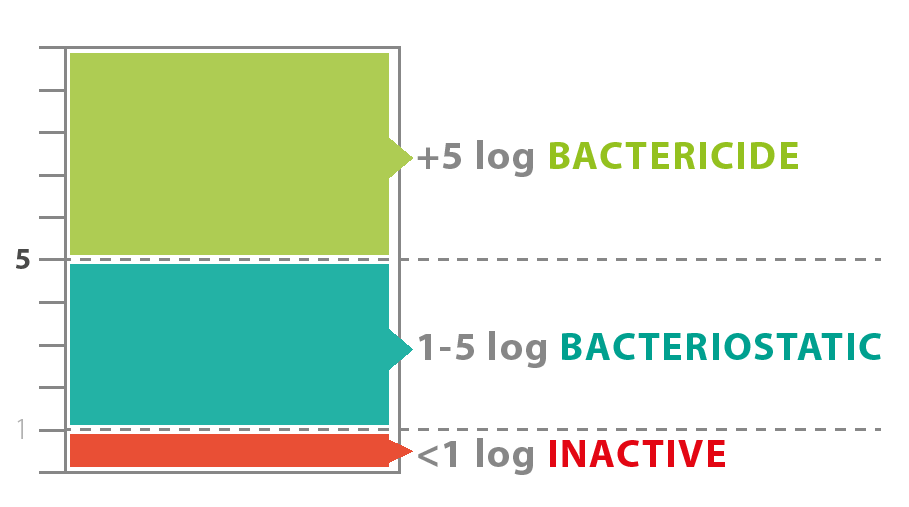
A plastic surface can only be called bactericide if more than 99% of all bacteria are eliminated. This is determined by the ISO 22196 test and classified according to EN 1040* standard.
ISO 22196 determines the bactericidal activity of a plastic surface by applying the following formula:
Bactericidal activity = log bacteria on control surface – log bacteria on treated surface
- Bactericide: when its bactericidal activity is 5 or higher, which translates to 100,000 times fewer bacteria on the treated surface than on the inactive control surface.
- Bacteriostatic: when its bactericidal activity is between 5 and 1 (100,000 to 10 times fewer bacteria).
- Inactive: when its bactericidal activity is lower than 1.
Tested to ISO 22196
![CS Acrovyn Bactericide Effectiveness - Escherichia coli[2020]](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-Escherichia-coli2020-200x143.png)
Activity in log: 5.4
Class: Bactericidal
![CS Acrovyn Bactericide Effectiveness - Staphylococcus aureus [2020]](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-Staphylococcus-aureus-2020-200x147.png)
Activity in log: 5.1
Class: Bactericidal
![CS Acrovyn Bactericide Effectiveness - Mycobacterium smegmatis [2020]](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-Mycobacterium-smegmatis-2020-200x147.png)
Activity in log: >5
Class: Bactericidal
![CS Acrovyn Bactericide Effectiveness - MRSA [2020]](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-MRSA-2020-200x136.png)
Activity in log: 3.92
Class: Bacteriostatic
![CS Acrovyn Bactericide Effectiveness - Salmonella enteritidis [2020]](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-Salmonella-enteritidis-2020-200x134.png)
Activity in log: 4.1
Class: Bacteriostatic
](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-Listeria-monocytogenes-20202-200x141.png)
Activity in log: 2.661
Class: Bacteriostatic
![CS Acrovyn Bactericide Effectiveness - Enterococcus species [2020]](https://c-sglobal.com/wp-content/uploads/2020/11/CS-Acrovyn-Bactericide-Effectiveness-Enterococcus-species-2020-200x138.png)
Activity in log: 2.28
Class: Bacteriostatic
* References:
- Sanitized® is a registered trademark of Sanitized AG.
- ISO 22196: 2011 ‘Measurement of antibacterial activity on plastics and other non-porous surfaces’
- EN 1040: 2005 ‘Chemical disinfectants and antiseptics – Quantitative suspension test for the evaluation of basic bacterial activity of chemical disinfectants and antiseptics – Test method and requirements (phase 1)’